About Whitsell Innovations, Inc.
Whitsell Innovations (WI), Inc. specializes in medical writing, quality control review, and electronic regulatory submissions. Since 2006, we have supported global pharmaceutical, biotech, and medical device clients with excellence in regulatory writing, publications, and safety documentation. Headquartered in Chapel Hill, North Carolina, our staff spans all US time zones and is composed of individuals with industry backgrounds and scientific educations, most with advanced degrees.
In 2019, we expanded our eCTD publishing services to support our clients with regulatory submission requirements, and in 2021 we invested substantially in the in-house development of tech-enablement tools to support our goal of perfect scientific, medical, and regulatory communication. In 2022, we were named the DA4S “8 That Innovate” runner up for our debut of our custom-programmed tools to further enhance our teams’ ability to exceed client expectations.
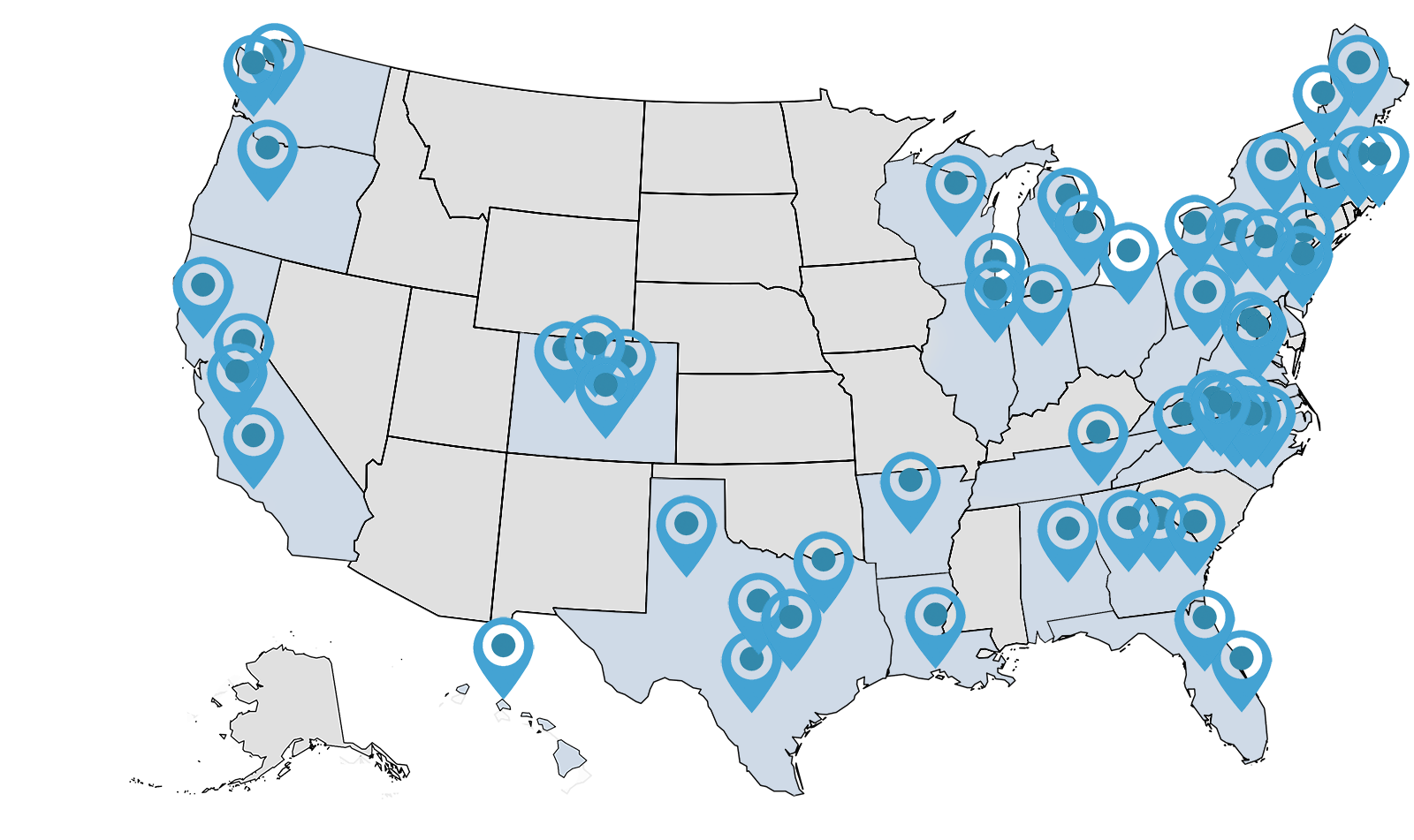
WI is a fully-remote company, with environmental, social, and corporate governance (ESG) core to our values. From the solar energy panels fueling our computer servers (at the home of our president) to hosting a school supplies drive for Kids in Need Foundation, we are committed to the well-being of the environment, our communities, our clients, our employees, and the patients we serve.
WI is a Women’s Business Enterprise National Council-certified, woman-owned small business.
We specialize in:
- Regulatory Writing
- Protocols
- Investigator’s Brochures
- Narratives
- Clinical Study Reports
- Chemistry, Manufacturing, and Control (CMC) Documentation
- Full Submissions (INDs, NDAs, 505(b)(2), 510ks, PMAs)
- Medical Communication
- Manuscripts and Abstracts
- Slides
- Sales Modules
- Patient and Physician Education
- Quality Control Review and Medical Editing
- Adherence to Style Guide
- Formatting
- Content (Abbreviation Checks, References)
- Data Verification
- eCTD Publishing
- Compilation
- Publishing
- Transmission to Health Authorities
As a boutique group, we offer a higher level of customer service and a commitment to individualized attention that larger and more diversified outsourcing organizations cannot provide.
WI caters to our clients’ needs, consulting on ways to improve processes, craft SOPs, and submit documents to federal regulatory agencies (including FDA and international submissions and NIH and SBIR grants). We help communicate complex corporate and scientific messages to lay audiences. Our singular focus is perfect scientific and medical communication directed at our client’s target audience. It’s what we do.
OUR MISSION
WI is a team of experts whose singular focus is perfect medical, scientific, and regulatory writing.
We support the life science community with clarity, reliability, and integrity.
We speak science, and we love what we do!
OUR PEOPLE
WI employs a 100% remote workforce. We are headquartered in Chapel Hill, North Carolina, and have folks in 27 states across the United States.